INTRODUCTION
End-Stage Renal Disease (ESRD) refers to kidney disease that has resulted in the permanent destruction of a sufficient number of nephron units that renal function (waste and/or water removal) must replaced using an artificial kidney (Renal Replacement Therapy). Chronic dialysis is provided either using Intermittent Hemodialysis (IHD) or Peritoneal Dialysis. Chronic IHD is usually provided 2-4 days per week (depending upon the type of renal dysfunction). Peritoneal dialysis can be provided as Continuous Ambulatory Peritoneal Dialysis (CAPD) or Continuous Cycling Peritoneal Dialysis (CCPD). In CAPD, patients can administer and manage passive exchanges 3-5 times per day. CCPD requires patients to connect to a machine (usually at night) for automated exchanges.
Peritoneal dialysis is rarely used in critical care. Patients who are receiving peritoneal dialysis who are stable may have PD continued in the critical care unit. It will be run by a dialysis nurse who is trained in PD. Most critically ill patients who need Renal Replacement Therapy (CRRT) will receive either IHD or Continuous Renal Replacement Therapy (CRRT). Both intermittent hemodialysis and continuous hemodialysis circuits utilize the same principles. Blood is removed from the patient, pumped through a dialysis filter and returned to the patient following removal of surplus water and wastes. The filter performs many of the functions of the kidney's nephron unit, hence, it is referred to as an "artificial kidney".
The major difference between intermittent and continuous therapies is the speed at which water and wastes are removed. Intermittent hemodialysis removes large amounts of water and wastes in a short period of time (usually over 2-4 hours), whereas, continuous renal replacement therapies remove water and wastes at a slow rate more consistent of that of native renal function. While intermittent dialysis allows chronic renal failure patients to limit the amount of time that they are connected to a machine, the rapid clearance of solutes and fluid can be poorly tolerated when a patient is hemodynamically unstable.
During an acute illness, patients with ESRD often require more frequent renal replacement therapy to manage their increased production of metabolic by-products. Patients who develop Acute Kidney Injury that does not resolve with shock management may also require acute renal replacement therapy. RRT for either group may be provided using either IHD or CRRT. Hemodynamic stability usually determines the method.
Intermittent hemodialysis and SLEDD are both delivered using a conventional hemodialysis machine that creates dialysis fluid (called dialysate) by adding electrolytes and salts to city water that has been dechlorinated and purified using reverse osmosis (RO). Dialysate fluid is not IV sterile, therefore, it cannot be delivered into the blood path. IHD and SLEDD require an IHD and an RO machine and are only run by IHD trained nurses at London Health Sciences Centre.
In CCTC, CRRT is provided using a Baxter PrisMaxTM or PrismaFlexTM machine. CRRT is delivered using sterile fluids, therefore, solutions can be delivered as either dialysis fluid or as replacement fluids into the blood path.
↑ Vissza a tartalomjegyzékhezACCESS
Historically, early dialysis circuits required the removal of blood from an artery with return of the "cleaned" blood to a vein. In the 1960's - 1970's, surgically implanted external arterial-to-venous shunts (e.g., Scribner, or Thomas shunts) were used for acute dialysis access. Early CRRT circuits required arterial and venous access devices (called Continuous Arterial-Venous circuits), as the arterial-venous blood pressure gradient was used to drive blood flow through the circuit. These arterial-venous CRRT circuits were fraught with challenges. Fluid removal could not be regulated , with effluent flow determined by the patency of the filter, the arterial-venous blood pressure gradient and the distance between the filter and the effluent collection bag. Over removal with hypotension often occurred at the start of treatment, with filter clotting imiting the duration of a treatment. With the introduction of a blood pump into the CRRT circuit, arterial-venous pressure gradients were no longer required for flow rates. This allowed temporary double lumen hemodialysis catheters to be introduced, eliminating the need for the surgical placement of arterial-to-venous shunts. This simplified initiation and lead to the development of the sophisticated technically available today. Although temporary circuits are all venous-venous today, red and blue labeling of catheter limbs and circuit tubing is universally used to identify the access limb (red is the usual side to remove blood) and return limb (blue denoting lumen where blood is returned).
Perm caths are double lumen dialysis catheters that are tunneled under the skin. They are made from material that is more appropriate for prolonged usage. Tunneling decreases the risk for infection or accidental dislodgement. The tunnelled portion of the catheter is separated from the external portion of the catheter by a dacron cuff which reduces migration of pathogens into the catheter tract and helps secure the device. Perm caths are usually inserted in Interventional Radiology Department. They are indicated for both prolonged dialysis in Acute Kidney Injury and ESRD.
ARTERIAL-VENOUS FISTULAS AND GRAFTS
Long term access for patients with ESRD may be created surgically through the use of an arterial-venous fistula or arterial-venous graft. Most are inserted in the upper limb, although, the lower limb can also be used.
An ARTERIAL-VENOUS FISTULA is the optimum access, created by suturing an artery and vein together. The capillary is bypassed, creating a high flow rate into the vein. This new circuit causes the vein to distend and bulge. It takes an average of 4 months before a fistula matures sufficiently to the point where it can be used for dialysis access. Consequently, for acute use, a temporary dialysis catheter may be required as a bridge until the fistula matures. At maturity, the fistula will change from being soft and pliable to firm and springy.
A buttonhole technique is typically used to access a fistula. Dull needles inserted repeatedly through the same hole will eventually cause a tunnelled tract, similar to an earing hole. Pain is minimized by using the same trac each time. The muscle layers of the artery close the tunnel after needle removal. Tunnel sites must be cleansed thoroughly and any scabs removed to prevent infection.
Fistulas are made from the patient's own biological material and are capable of functioning for many years. They are completely under the skin, which reduces infection when compared to double lumen catheters, and allows patients to swim and submerge in water. The high flow rates also reduce clotting
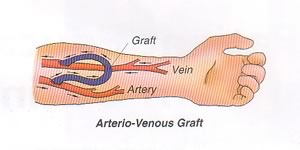
An ARTERIAL-VENOUS GRAFT is another surgical option, used when the vessels are too small to support the formation of a fistula. Graft material is used to create a conduit between the artery and vein. A graft requires less time to mature than a fistula, and may be accessed as early as 4 weeks after insertion. It is also easier to create. A graft is also associated with a lower rate of infection than a central venous catheter and allows the patient to swim.
Compared to fistulas, grafts do not survive as long but they are easier to implant. They also require needle access each time (fistulas do not generate tunneled tracks). Grafts are also more likely to clot.
Complications of fistulas or grafts include:
Infection
Clotting
Aneurysm (risk increases if the same tunnel cannot be used each time or if sharp needles are required)
Stenosis (arterial of venous)
Steal phenomena" (arterial flow being "stollen" from one area to another to "feed" the shunt).
ASSESSMENT AND MONITORING
Once mature, both fistulas and grafts should maintain a high rate of blood flow. This can be assessed by the following:
THRILL: A thrill is an "electrical sensation" or tingling created by the rapid flow of blood through the circuit. It can be felt by gently touching over top of the fistula.
BRUIT: A bruit is an audible whooshing sound that is heard when a stethoscope is placed gently over the fistula or shunt. This is created by the rapid flow of blood.
MONITOR: Watch for signs of complications including swelling at the site or distal extremity, redness or discharge.
AVOID: Avoid taking blood pressure, applying a tourniquet or constrictive clothing or excessive use of the affected limb.
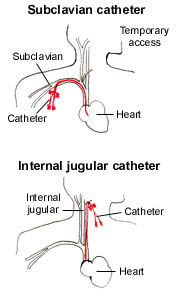
In critical care, temporary double-lumen venous dialysis catheters are the most common form of access. They can be inserted quickly at the bedside and used immediately. "Perm" catheters are double lumen venous catheters that are designed for longer indwelling use. They are used more frequently in patients with chronic renal failure and may be used as a bridge until a surgically created fistula is ready for use.
Dialysis catheters are easy to differentiate from regular intravenous lines by their red and blue hubs. The red lumen denotes the side of the venous catheter that is used to pull blood from the patient, and is referred to as the access lumen. The blue lumen is the return site and is used to reinfuse the patient's blood after it passes through the dialysis filter. If an adequate flow rate cannot be achieved by removing blood from the access side of a catheter, the catheter limbs can be reversed during dialysis. Reversal of the limbs does produce a small reduction in clearance due to recirculation that is not usually clinically important.
When a double lumen catheter is not in use for dialysis, some form of anticoagulant is always instilled into each lumen to maintain patency. While citrate is the most common agent, some catheters may still require blocking with heparin or tPA (particularly among patients with ESRD). If heparin is used, the concentration may be as high as 5,000 - 10,000 units per mL. Because each lumen contains a volume of ~1 to 2 ml, the two lumens could contain a maximum of up to 40,000 units of heparin! ALWAYS assume that each lumen contains full strength heparin (even if it is labeled as containing saline).
In CCTC, 4% Citrate is now the standard catheter blocking solution for dialysis catheters. Citrate binds to calcium to prevent clotting and does not affect the aPTT. Citrate is the standard for blocking all CRRT catheters in CCTC, even when heparin has been used to maintain filter patency.
EMERGENCY VASCULAR ACCESS:
A double-lumen venous dialysis catheter can be used as a central venous infusion site during an emergency. To ensure the line remains patent for subsequent dialysis treatments and to reduce the risk for infection, these lines should only be used for dialysis.
If a dialysis catheter is the only vascular access available in a life-threatening emergency, it can be used as a central line. HOWEVER, always assume that the catheter contains heparin, TPA or another agent. Aspirate at least 5 ml from each lumen and flush vigorously before connecting to an infusion pump to maintain patency. Maintain sterility to preserve the site for future access.
↑ Vissza a tartalomjegyzékhezHemodialysis employs the principles of diffusion, hemofiltration and convection, using an external filter to create an artificial nephron unit.
Recall the normal nephron unit:
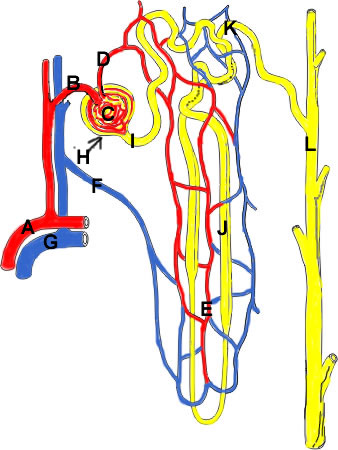
|
↑ Vissza a tartalomjegyzékhez
4.
ARTIFICIAL KIDNEY
The dialysis filter is referred to as an artificial kidney. Blood is pulled from the patient and carried into the filter. Once inside, the blood travels through many tiny tubules called hollow fibers. Water and solutes can pass across the semi-permeable membrane between the blood and the fluid that surrounds the hollow fibers. Any fluid or solutes that enters the filter canister will be drained out as waste. 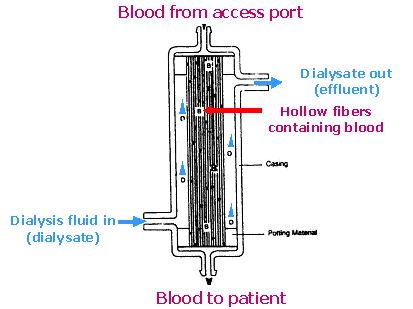 Schematic of dialysis filter (artificial kidney) Note how the dialysis filter has structural similarities to the nephron unit. Blood arrives at the filter via the access tubing (afferent arteriole). Blood enters the small hollow fibers within the filter (glomerulus). Water and solutes diffuse across the semi-permeable membrane of the hollow fibers and collect in the canister (Bowman's Capsule). Collected fluid (filtrate or effluent) is then removed via the drainage tubing (collecting tubule). Blood that remains in the hollow fibers is returned to the patient via the return side of the filter (efferent arterial).
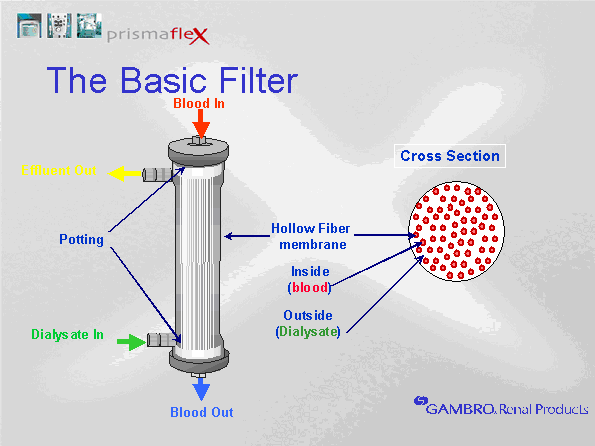 Although similarities exist between the nephron unit and the artificial kidney, the artificial kidney has limited capabilities. In the nephron unit, filtered water and waste enters the proximal tubule. Because the nephron unit removes significantly more water and solutes than needed, most of the water and electrolytes that enter the tubule system are reabsorbed. Unlike the nephron unit, the artificial kidney cannot reabsorb water or solutes that enter the filter canister Any filtrate that enters the filter canister will be removed via the drainage tubule. Consequently, one of the differences in the artificial kidney is the absence of the proximal tubule, Loop of Henle and distal tubule where water and solute reabsorption and secretion occurs. Thus, the drainage tubule that exits the filter is similar to the collecting tubule of the nephron unit, not the proximal tubule. To compensate for the inability to reabsorb water and solutes following removal from the blood, the artificial kidney is manipulated to restrict the actual removal to only surplus water and wastes. This is done by adjusting dialysis solutions and ultrafiltration rates. If more water or solutes are removed than desired, they may need to be given back via intravenous infusions. The artificial kidney does not replace other important kidney functions, including stimulation of red blood cell production (erythropoietin), blood pressure and sodium regulation (renin) and calcium uptake by the GI tract (vitamin D synthesis). The nephron normally traps and recycles bicarbonate to maintain acid base balance. Bicarb is given to patients during hemodialysis to compensate for bicarb deficits. The principles used during hemodialysis are reviewed below: ↑ Vissza a tartalomjegyzékhez
5.
DIFFUSION
Diffusion is the movement of particles (solutes) across a semi-permeable membrane. Diffusion is the movement f 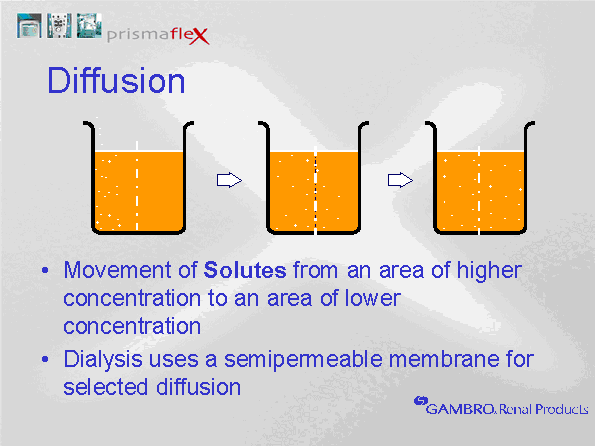 From the side with the highest concentration of particles, to the side with the lowest concentration. ↑ Vissza a tartalomjegyzékhez
6.
DIALYSIS FLUID (DIALYSATE):
Dialysate is the fluid that is pumped into the filter canister, surrounding the hollow fibers. The concentration of solutes in the dialysis fluid determines diffusion gradients. The removal of surplus solutes from the blood is achieved by infusing dialysate fluid that contains a lower solute concentration than the serum concentration (e.g. dialysate does not contain urea or creatinine). To maintain normal serum electrolyte levels, dialysate fluid contains sodium, chloride and magnesium levels that are equal to serum concentrations (thus, removal of these electrolytes should only occur if the blood level exceeds normal serum concentrations). In renal failure, potassium is often high at the start of a treatment, therefore, we may begin dialysis with a low concentration of potassium in the dialysate. Because potassium is easily removed during dialysis, and continued dialysis will be required to ensure removal of other wastes such as urea and creatinine, potassium concentrations in the dialysate often require upward adjustment as the potassium level in the blood falls. Although in theory, potassium levels should not fall below 4 mmol/L in the serum if the dialysate contains 4 mmol/L, a number of factors influence serum potassium levels in critical care. Insulin therapy and the use of sympathomimetic drugs promotes the movement of potassium from the blood into the cells. This can lower serum levels. Additionally, potassium loss through the GI tract can increase the potential for hypokalemia. Low magnesium levels will also suppress the serum potassium levels, therefore, magnesium deficits should be replaced as needed. Additionally, high hemofiltration rates can lead to additional potassium clearance. Potassium levels must be monitored closely and adjusted to maintain normal serum concentrations. In renal failure, serum bicarbonate levels are generally low, therefore, a source of bicarbonate is added to the dialysate to facilitate diffusion of bicarbonate into the blood. At one time, lactate based products were used for CRRT. Lactate is metabolized to bicarbonate to provide a less expensive source of bicarbonate replacement with longer product stability. Because critically ill patients with organ dysfuncition will often have impaired hepatic clearance, lactate based products were frequently associated with prolonged lactate elevation. We no longer use lactate-based products. All dialysis solutions that are currently in use in CCTC are now bicarbonate based. We use 3 primary solutions, all contain 32 mmol/L of bicarbonate after mixing. The bicarbonate is contained in an upper compartment of a 5 L dialysis bag. To release the bicarbonate, squeeze the upper compartment until the two compartments mix. Failure to mix the two compartments can result in severe electrolyte abnormalities. The contents in compartment A, B and A plus B are listed on each bag. As well, failure to mix the two compartments would mean that only the volume in the lower compartment would be accessible. The scale would still weigh the bag and attempt to "draw" the upper compartment volume, pulling air into the circuit. Dialysis fluid does not in theory cross into the blood side of the filter. It runs along the outside of the blood filter and into the effluent drainage. Whatever dialysis volume is administered, an equal volume will be removed in the effluent. This is automatic (dialysate input = dialysate output). Recall that hemodialysis solution is not IV sterile in IHD. For this reason, some fluid removal must always be maintained to prevent any crossing of dialysis fluid to the blood side. In CRRT, solutions are IV sterile. Therefore, patient fluid removal can be turned to zero because there is no risk of infection should dialysis fluid enter the blood path. When patient fluid removal is set to zero, the hourly fluid removal might be slightly positive (usually 1-5 ml per hour). This actually indicates a gain of patient fluid by 1-5 ml. Concentration gradients play a major role in diffusion. These will be explored further in the discussion on clearance. The other factor that influences diffusion is the type of filter used and the size of the molecule to be cleared. Diffusion of solutes cannot occur across a concentration gradient if the pore size is too small to permit passage. 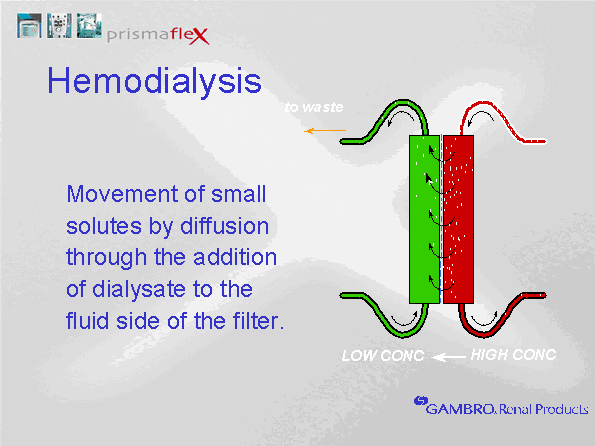 ↑ Vissza a tartalomjegyzékhez
7.
ULTRAFILTRATION
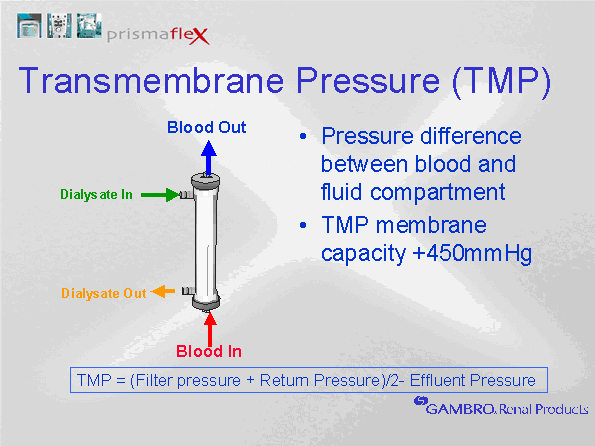
Ultrafiltration is the movement of water across a semi-permeable membrane because of a pressure gradient (hydrostatic, osmotic or oncotic). The increased blood pressure in the glomerulus creates a favourable driving pressure to force water across the glomerular membrane. Blood pressure within the hollow fibers is positive, while the pressure outside the hollow fibers is lower. Increased negativity can be generated outside the hollow fibers by the effluent pump by either increasing the fluid removal rate, or by increasing the replacement flow rate. The difference between the blood pressure in the hollow fibers and the surrounding pressure is the Transmembrane Pressure (TMP). The TMP determines the ultrafiltrate production. Different filter membrane properties can produce different ultrafiltration rates at a constant TMP. A filter that is more permeable to water will allow more water to travel across the membrane at a given TMP. A filter with a high permeability to water is called a high flux membrane. 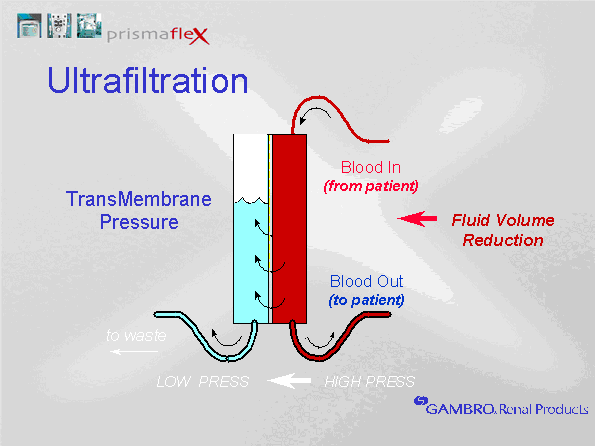 ↑ Vissza a tartalomjegyzékhez
8.
HEMOFILTRATION
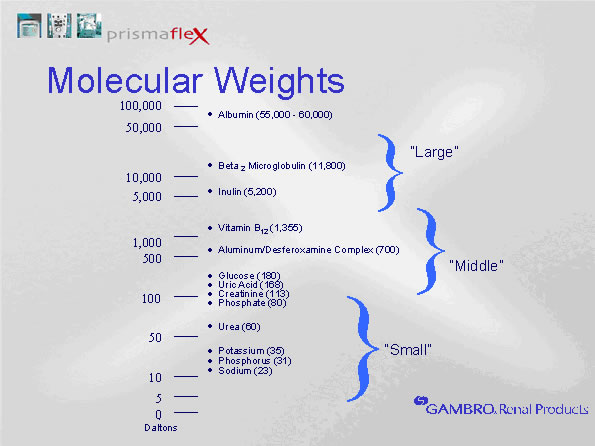
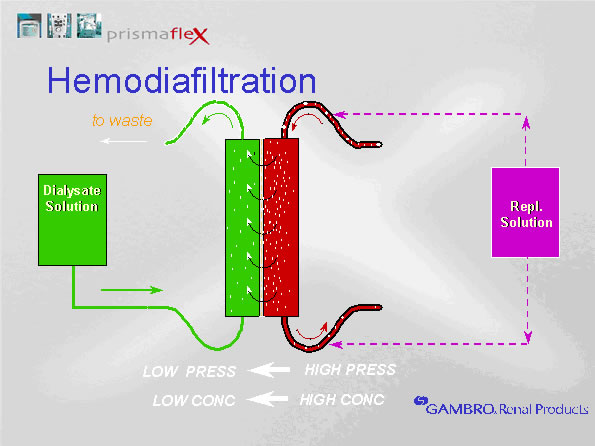
In hemodialysis circuits, pulling large volumes of water across the semi-permeable membrane creates a convective current that "drags" additional solutes. While diffusion is effective at removing most small molecules, convection enhances the removal of small and mid-sized molecules. Thus, convection can be added to hemodialysis therapy to enhance solute removal. To prevent hypovolemia, any water removed during hemofiltration must be returned to the blood before it reaches the patient. This is called "replacement" fluid. Hemofiltration rates of 1 L/hr mean that one liter of fluid is removed from the patient's blood and eliminated in the drainage fluid AND 1 L of replacement fluid is returned to the circuit before it reaches the patient. We set hemofiltration rates by adjusting replacement rates. Any fluid removed during hemofiltration is given back to maintain a net neutral fluid balance. Replacement fluid must be sterile intravenous fluids with concentrations of electrolytes similar to plasma. For example, if the CRRT therapy includes a hemofiltration rate of 1 L per hour, and the fluid removal is set at 200 ml per hour, 1200 ml will be pulled from the patient and introduced into the drainage collection bag each hour. Because the 1 L of hemofiltration is replaced, the net fluid removed is 200 ml. Whether hemofiltration is used or not, the net fluid removed is equal to the fluid removal setting.  ↑ Vissza a tartalomjegyzékhez
9.
PREDILUTION VERSUS POSTDILUTION HEMOFILTRATION
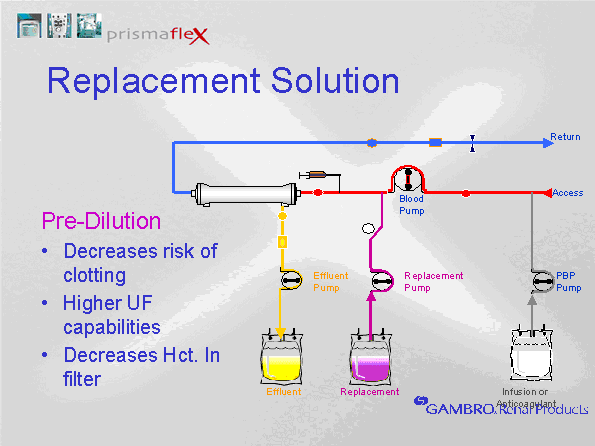
PREDILUTION VERSUS POSTDILUTION HEMOFILTRATION Replacement fluids can be returned either pre or post filter. This is referred to as predilution or post dilution sets. Predilution means that the replacement solution is returned to the blood before it reaches the filter, diluting the blood in the hollow fibers. Postdilution means that the replacement fluid is returned to the blood after the filter (but before the return side of the access catheter). Predilution dilutes the blood in the filter, reducing clotting. Postdilution concentrates the blood in the filter, enhancing clearance.
|